This is the story of why we are building Endura. Other articles will take a deeper dive into the chemistry and technology.
The genomic disease map is remarkably detailed
Two decades ago, we sequenced the human genome and, for the first time, could read the tome of human biology. Today, we've found the typos, but we still can't fix most of them.
Human genomics has indicted specific culprits, from the inherited mutations that cause thousands of rare diseases to the somatic changes that accumulate over a lifetime to drive cancer and neurodegeneration. A child born with spinal muscular atrophy (SMA) is missing a working copy of the SMN1 gene, causing their motor neurons to progressively die until they lose the musculature to breathe. An adult's cancer can often be traced back to specific driver genes that, when dysregulated, fuel the tumor's unchecked growth.

Drug developers have historically worked downstream, targeting not the gene itself but the protein it produces, finding a molecule that binds and inhibits the disease-causing protein. That enormously successful approach has produced nearly all of the most important drugs in history. But we've largely addressed the proteins that cooperate; what remains are the ones with no good binding site, no handle, no place for a drug to grab hold. Despite decades of effort, these undruggable targets persist, and they include some of the most important disease drivers that genomics has identified.[1]
Gene-targeting medicines work, but are limited by delivery
Over the past two decades, scientists built an entirely new class of medicines that go upstream of the protein to target the genetic message directly. ASOs, siRNAs, CRISPR, and gene therapies can reach into a cell and silence an mRNA or edit the DNA itself. The results have been extraordinary.
Before Spinraza, the first ASO approved for SMA, parents watched their children lose the ability to move and breathe, knowing most would not live to see their second birthday. Now, children who would have died are sitting up and walking. Before the first CRISPR therapy for sickle cell disease, patients suffered recurring crises of excruciating pain as misshapen blood cells blocked their vessels. Now, after a single treatment, many are pain-free for the first time. The science of drugging the genetic message is no longer theoretical.
But these medicines share a fundamental physical limitation: they are large molecules that don’t readily cross cell membranes and can only reach a handful of the body's tissues. Delivery to the liver has been solved by GalNAc, delivery to the brain is achievable at the cost of a needle into the spinal canal, and delivery to muscle is now possible with antibody conjugation.[2] But this leaves us without solutions for most organs, including heart, lung, and kidney, as well as almost all solid tumors. The number of disease-causing genes that no medicine can reach remains enormous.
.png)
Small molecules can target the genetic message
The field has been working to solve this delivery challenge tissue by tissue, engineering new targeting strategies for each organ. But rather than delivering large molecules past barriers they weren't designed to cross, what if we empowered small molecules, which already distribute throughout the body, to target the genetic message directly?
This is possible. Roche developed a small molecule drug for SMA, called Evrysdi, that works by modifying RNA splicing. It has steadily overtaken the incumbent genetic medicines since its launch, because patients and families chose a medicine they could take by mouth, at home, every day, over an ASO requiring repeated spinal injections or a $2,000,000 gene therapy.
.png)
Evrysdi demonstrated that a small molecule can achieve genetic precision through RNA. This splice modulation has generalized to several more drugs in clinical trials for other diseases. But the approach is inherently constrained because it works by redirecting the cell's splicing machinery at specific splice sites, which represent < 1% of any given mRNA. Most of the sequence is still inaccessible.
Endura takes a different approach. Our molecules use covalent chemistry to scar an mRNA, physically blocking the ribosome from making the disease-causing protein. Because this mechanism isn’t constrained by splicing biology, binding nearly anywhere in the mRNA is productive, expanding the targeting real estate by > 50-fold. It is gene-level precision like CRISPR, but in a pill.
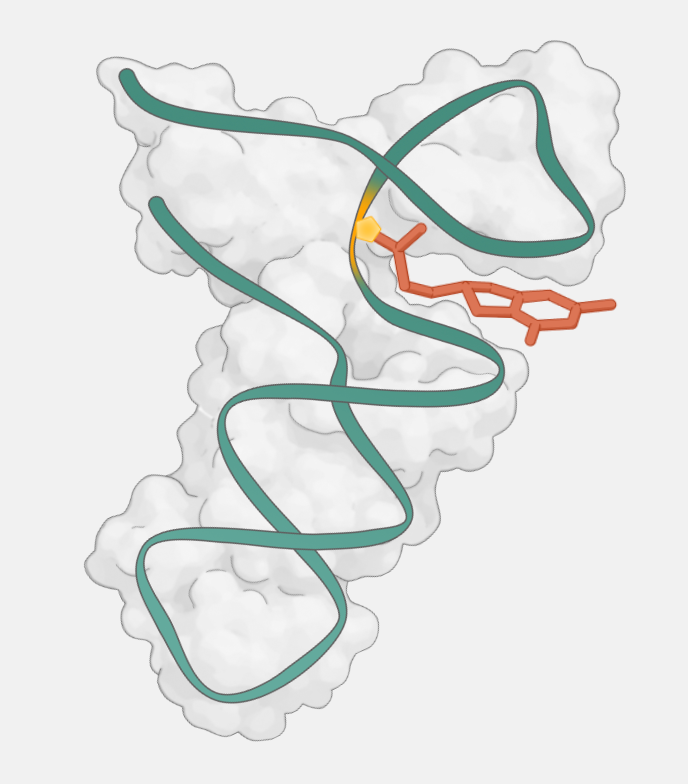
RNA has resisted small molecule drug discovery for years, and for real reasons: the targets are flexible, transient, and structurally repetitive in ways that make selective binding exceptionally difficult. But two advances have changed the calculus. First, breakthroughs in covalent chemistry allow us to bring a selective, functionally disruptive modification to RNA for the first time. Second, massively scalable sequencing lets us screen drugs against all RNA targets simultaneously, turning drug discovery from a one-target-at-a-time effort into a systematic search.
We imagine a future where drugging the genetic message is as commonplace as drugging the protein, where the thousands of undrugged disease drivers that genomics identified become accessible to oral medicines. That would complete the genomics revolution, and it's what we're building toward.
A medicine's value is how many people it can help
If the goal is to target the genetic message, why build a small molecule company rather than better delivery for genetic medicines? Our answer comes down to what happens after the science works.
The pharmaceutical industry operates on a social contract: companies earn returns during a patent-protected period, and then generics make the medicine accessible to everyone. This is what brought HIV treatment from over $10,000 to under $100 per year in sub-Saharan Africa, because the drugs were small molecule pills produced for pennies per dose and shipped anywhere without refrigeration. It breaks when the medicine requires expensive manufacturing or administration by a healthcare professional.[3] Many modalities have a structural floor on cost and complexity that no amount of optimization can break through, but a small molecule pill doesn't have that floor.
We chose to build a company around RNA-targeting small molecules not just because of what they can do, but because of who they can reach at the end of the day.

